Information
28 Namyang-ro 930beon-gil, Namyang-eup, Hwaseong-si, Gyeonggi-do, South Korea
+82 31-357-2610
-
Overview
- Summary
- DONGIL TECHNOLOGY, established in 1986, is a specialized manufacturer of medical devices and electronic components. The company has grown based on its EMI/EMC filter business, primarily producing ultrasonic surgical devices and scalers for medical use, leveraging its piezoelectric ceramic application technology. It is also expanding its business into various industrial sectors, including environmental components, industrial products, and vision inspection solutions.DONGIL TECHNOLOGY, established in 1986, is a specialized manufacturer of medical devices and electronic components. The company has grown based on its EMI/EMC filter business, primarily producing ultrasonic surgical devices and scalers for medical use, leveraging its piezoelectric ceramic application technology. It is also expanding its business into various industrial sectors, including environmental components, industrial products, and vision inspection solutions.
- Key Products/Technologies
- Sonic Surgeon 600L: An ultrasonic surgical device offering high output and enhanced patient safety. It features an operating frequency of 23-29Khz and a maximum water supply of 100ml/min to minimize bone heating. The product allows for the application of 22 different tips, including a patented crestal approach tip, for various dental surgeries.Sonic Surgeon 600L: An ultrasonic surgical device offering high output and enhanced patient safety. It features an operating frequency of 23-29Khz and a maximum water supply of 100ml/min to minimize bone heating. The product allows for the application of 22 different tips, including a patented crestal approach tip, for various dental surgeries.
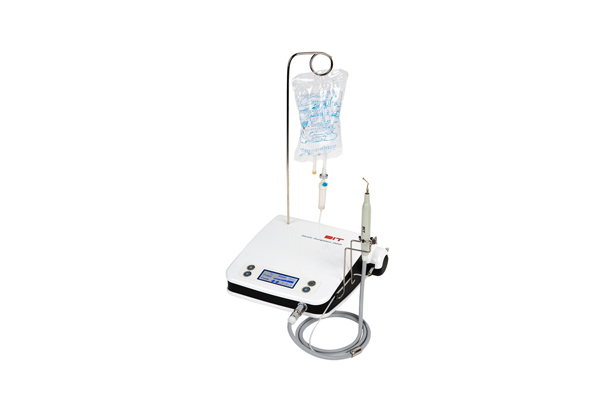
- Sonic Surgeon 310L: An ultrasonic surgical device characterized by a bright LED handpiece and a colored touch LCD. It has an operating frequency of 23-29Khz and dimensions of 300*100*300mm. Based on its proprietary piezoelectric ceramic and circuit technology, it provides stable output and features a water supply function to prevent bone heating.Sonic Surgeon 310L: An ultrasonic surgical device characterized by a bright LED handpiece and a colored touch LCD. It has an operating frequency of 23-29Khz and dimensions of 300*100*300mm. Based on its proprietary piezoelectric ceramic and circuit technology, it provides stable output and features a water supply function to prevent bone heating.
- Sonic Surgeon 300: A powerful ultrasonic surgical device with various tips, having obtained international approvals such as CE, FDA, MHWL, and GOST. It boasts stable and powerful output through proprietary piezo technology, and a 100ml/min water infusion feature minimizes bone heating. The ergonomic foot switch design ensures convenient operation.Sonic Surgeon 300: A powerful ultrasonic surgical device with various tips, having obtained international approvals such as CE, FDA, MHWL, and GOST. It boasts stable and powerful output through proprietary piezo technology, and a 100ml/min water infusion feature minimizes bone heating. The ergonomic foot switch design ensures convenient operation.
- Sonic Buff 310L: An ultrasonic scaler optimally installable within a dental unit chair. It features 6 LED lights and a grip button on the handpiece for adjustable light intensity, enhancing user convenience with its ergonomic design. The product offers stable ultrasonic power and excellent durability, with hygienic autoclavable sterilization at 135℃.Sonic Buff 310L: An ultrasonic scaler optimally installable within a dental unit chair. It features 6 LED lights and a grip button on the handpiece for adjustable light intensity, enhancing user convenience with its ergonomic design. The product offers stable ultrasonic power and excellent durability, with hygienic autoclavable sterilization at 135℃.
- Sonic Buff 300: A standard ultrasonic scaler for various applications. Used for dental plaque removal and alveolar bone treatment, it optimizes frequency according to the attached tip for safe and effective procedures. It provides a spacious sterilization tray capable of sterilizing the handpiece, torque wrench, and tips together at 135℃.Sonic Buff 300: A standard ultrasonic scaler for various applications. Used for dental plaque removal and alveolar bone treatment, it optimizes frequency according to the attached tip for safe and effective procedures. It provides a spacious sterilization tray capable of sterilizing the handpiece, torque wrench, and tips together at 135℃.
- EMI/EMC Solutions: Electromagnetic interference (EMI) filter technology applied to industrial, household, and defense electronic devices for noise suppression. The company is one of the leading domestic firms in this field, possessing global competitive technology.EMI/EMC Solutions: Electromagnetic interference (EMI) filter technology applied to industrial, household, and defense electronic devices for noise suppression. The company is one of the leading domestic firms in this field, possessing global competitive technology.
- Piezoelectric Ceramic Application Technology: Proprietary piezoelectric ceramic material technology applied to high-precision piezoelectric actuators and core medical device components. This core capability enables stable and powerful output for medical ultrasonic surgical devices.Piezoelectric Ceramic Application Technology: Proprietary piezoelectric ceramic material technology applied to high-precision piezoelectric actuators and core medical device components. This core capability enables stable and powerful output for medical ultrasonic surgical devices.
- Core Advantages
- Over 30 Years of Experience and Accumulated Technical Know-how: Extensive market experience and technical expertise gained through dedicated development of electronic components and products since its establishment in 1986.Over 30 Years of Experience and Accumulated Technical Know-how: Extensive market experience and technical expertise gained through dedicated development of electronic components and products since its establishment in 1986.
- Strong R&D Expertise and Proprietary Piezoelectric Ceramic Technology: A robust R&D capability with one-third of its employees in research, holding top-tier piezoelectric ceramic application technology domestically and internationally. Vertical integration capability for in-house production of core medical device components.Strong R&D Expertise and Proprietary Piezoelectric Ceramic Technology: A robust R&D capability with one-third of its employees in research, holding top-tier piezoelectric ceramic application technology domestically and internationally. Vertical integration capability for in-house production of core medical device components.
- Category Leader in EMI/EMC Filter Sector: One of the leading domestic companies in the EMI/EMC filter business for industrial, household, and defense electronic devices, possessing global competitiveness.Category Leader in EMI/EMC Filter Sector: One of the leading domestic companies in the EMI/EMC filter business for industrial, household, and defense electronic devices, possessing global competitiveness.
- Customer-Centric Solution Provision and Trust Relationship: A strong trust relationship built through mobilizing technical capabilities and cooperation to find suitable solutions for customers. Deep understanding of customer needs and the ability to develop customized products.Customer-Centric Solution Provision and Trust Relationship: A strong trust relationship built through mobilizing technical capabilities and cooperation to find suitable solutions for customers. Deep understanding of customer needs and the ability to develop customized products.
- Stable Financial Structure and Continuous Investment: Maintaining a sound financial statement through debt-free management and actively investing in new product R&D and advanced production lines, positioning it as a stable partner.Stable Financial Structure and Continuous Investment: Maintaining a sound financial statement through debt-free management and actively investing in new product R&D and advanced production lines, positioning it as a stable partner.
- International Medical Device Certifications: Major medical device products like Sonic Surgeon 300, 310L, and 600L have obtained international certifications such as CE, FDA, MHWL, and GOST, ensuring product reliability and market access.International Medical Device Certifications: Major medical device products like Sonic Surgeon 300, 310L, and 600L have obtained international certifications such as CE, FDA, MHWL, and GOST, ensuring product reliability and market access.
- Target Industries
- Medical Device Industry: Dental treatment fields such as implant surgery, oral surgery, periodontal surgery, maxillofacial surgery, scaling, and teeth whitening, through its dental ultrasonic surgical devices (Sonic Surgeon series) and scalers (Sonic Buff series).Medical Device Industry: Dental treatment fields such as implant surgery, oral surgery, periodontal surgery, maxillofacial surgery, scaling, and teeth whitening, through its dental ultrasonic surgical devices (Sonic Surgeon series) and scalers (Sonic Buff series).
- Electronic Components Industry: Providing EMI/EMC filter solutions to industrial, household, and defense electronic devices for electromagnetic noise suppression.Electronic Components Industry: Providing EMI/EMC filter solutions to industrial, household, and defense electronic devices for electromagnetic noise suppression.
- Semiconductor and Display Industry: Supplying components utilizing piezoelectric ceramic material technology, such as high-precision piezoelectric actuators.Semiconductor and Display Industry: Supplying components utilizing piezoelectric ceramic material technology, such as high-precision piezoelectric actuators.
- Environmental Industry: Offering environmental components and products for atmospheric environment monitoring, removal of airborne foreign substances, and purification.Environmental Industry: Offering environmental components and products for atmospheric environment monitoring, removal of airborne foreign substances, and purification.
- Manufacturing Industry: Contributing to yield management and productivity enhancement in manufacturing sites through its AI vision and collaborative robot-based vision inspection solution 'inno-eye'.Manufacturing Industry: Contributing to yield management and productivity enhancement in manufacturing sites through its AI vision and collaborative robot-based vision inspection solution 'inno-eye'.
- Major Markets
- South KoreaSouth Korea
- Certifications/Patents
- CE Certification: European Union conformity certification for major medical device products such as Sonic Surgeon 300, Sonic Surgeon 310L, and Sonic Surgeon 600L.CE Certification: European Union conformity certification for major medical device products such as Sonic Surgeon 300, Sonic Surgeon 310L, and Sonic Surgeon 600L.
- FDA Approval: U.S. Food and Drug Administration approval for the Sonic Surgeon 300 product.FDA Approval: U.S. Food and Drug Administration approval for the Sonic Surgeon 300 product.
- MHWL Approval: Ministry of Health, Labour and Welfare (Japan) approval for the Sonic Surgeon 300 product.MHWL Approval: Ministry of Health, Labour and Welfare (Japan) approval for the Sonic Surgeon 300 product.
- GOST Approval: Russian State Standard approval for the Sonic Surgeon 300 product.GOST Approval: Russian State Standard approval for the Sonic Surgeon 300 product.
- ISO 13485: International standard certification for medical device quality management systems.ISO 13485: International standard certification for medical device quality management systems.
- GMP (Good Manufacturing Practice): Compliance with medical device manufacturing and quality control standards.GMP (Good Manufacturing Practice): Compliance with medical device manufacturing and quality control standards.
- Patented Crestal Approach Tip: A patented technology applied to the Sonic Surgeon series, enabling safe and convenient sinus surgery.Patented Crestal Approach Tip: A patented technology applied to the Sonic Surgeon series, enabling safe and convenient sinus surgery.
- Patent Pending for Handpiece LED Illumination Intensity Control: A technology applied to Sonic Buff 310L, allowing LED illumination intensity adjustment via a grip button on the handpiece, currently patent pending.Patent Pending for Handpiece LED Illumination Intensity Control: A technology applied to Sonic Buff 310L, allowing LED illumination intensity adjustment via a grip button on the handpiece, currently patent pending.
Introduction
Key Products
Location
28 Namyang-ro 930beon-gil, Namyang-eup, Hwaseong-si, Gyeonggi-do, South Korea
클릭하여 위치 살펴보기